Publications
Please see my Google Scholar page for the most up-to-date list
* equal contributions
2025
-
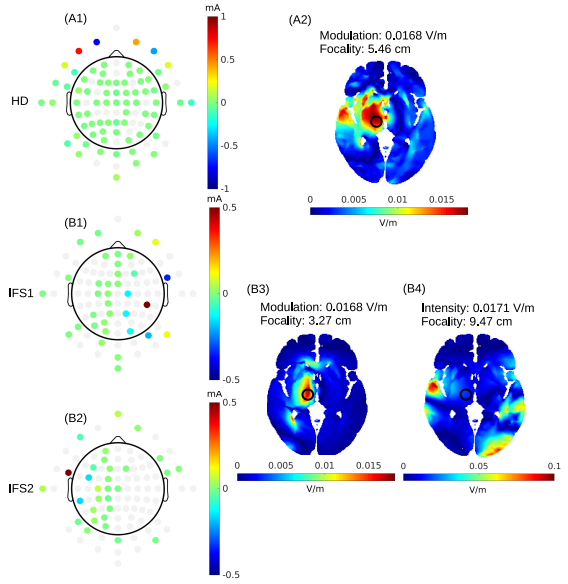
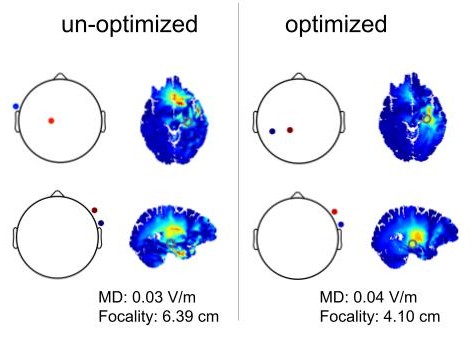 On the need of individually optimizing temporal interference stimulation of human brains due to inter-individual variabilityTapasi Brahma, Alexander Guillen, Jeffrey Moreno, Abhishek Datta, and Yu HuangBrain Stimulation, 2025
On the need of individually optimizing temporal interference stimulation of human brains due to inter-individual variabilityTapasi Brahma, Alexander Guillen, Jeffrey Moreno, Abhishek Datta, and Yu HuangBrain Stimulation, 2025Introduction Transcranial temporal interference stimulation (TI, TIS, or tTIS), also known as interferential stimulation (IFS), is able to focally stimulate deep brain regions, provided it is properly optimized. We previously presented an algorithm for optimizing TI using two arrays of electrodes and showed that it can achieve more focal stimulation compared to optimized high-definition transcranial electrical stimulation (HD-TES) and conventional optimized TI using two pairs of electrodes, especially in the deep brain areas such as the hippocampus. However, those modeling studies were only performed on an averaged head (MNI152 template) and three individual heads without exploring inter-individual variability. Existing TI works in the literature mostly utilize a common (possibly optimized) montage of two pairs of electrodes on different individual heads without considering inter-individual variability. Material and method: Here we aim to study the inter-individual variability of optimized TI by applying the same optimization algorithms on N = 25 heads using their individualized head models. Specifically, we compared the focality achieved by different stimulation techniques at six different regions of interest (ROI; right hippocampus, left dorsolateral prefrontal cortex, left motor cortex, right amygdala, right caudate, and left thalamus) under both individually optimized and unoptimized montages. We also conducted numerical sensitivity analysis on the individual optimization and performed phantom recordings to test our models. Results As expected, there is a variability in focality achieved by TI of up to 1.2 cm at the same ROI across subjects due to inter-individual differences in the head anatomy and tissue conductivity. We show that optimized TI using two arrays of electrodes achieves higher focality than that from optimized HD-TES at the same level of modulation intensity at 5 of the 6 ROIs. Compared to using a common montage either optimized from the MNI152 template or from the literature, individually optimized TI using two pairs of electrodes improves the focality by up to 4.4 cm, and by up to 1.1 cm if using two arrays of electrodes. Focality achieved by the individual optimization is sensitive to random changes and can vary up to 9.3 cm due to the non-lienarity of TI physics. Experimental recordings on a head phantom confirms the drop in TI stimulation strength when using unoptimized montages as predicted by our in silico models. Conclusion This work demonstrates the need of individually optimizing TI to target deep brain areas, and advocates against using a common head model and montage for TI modeling and experimental studies.
2024
-
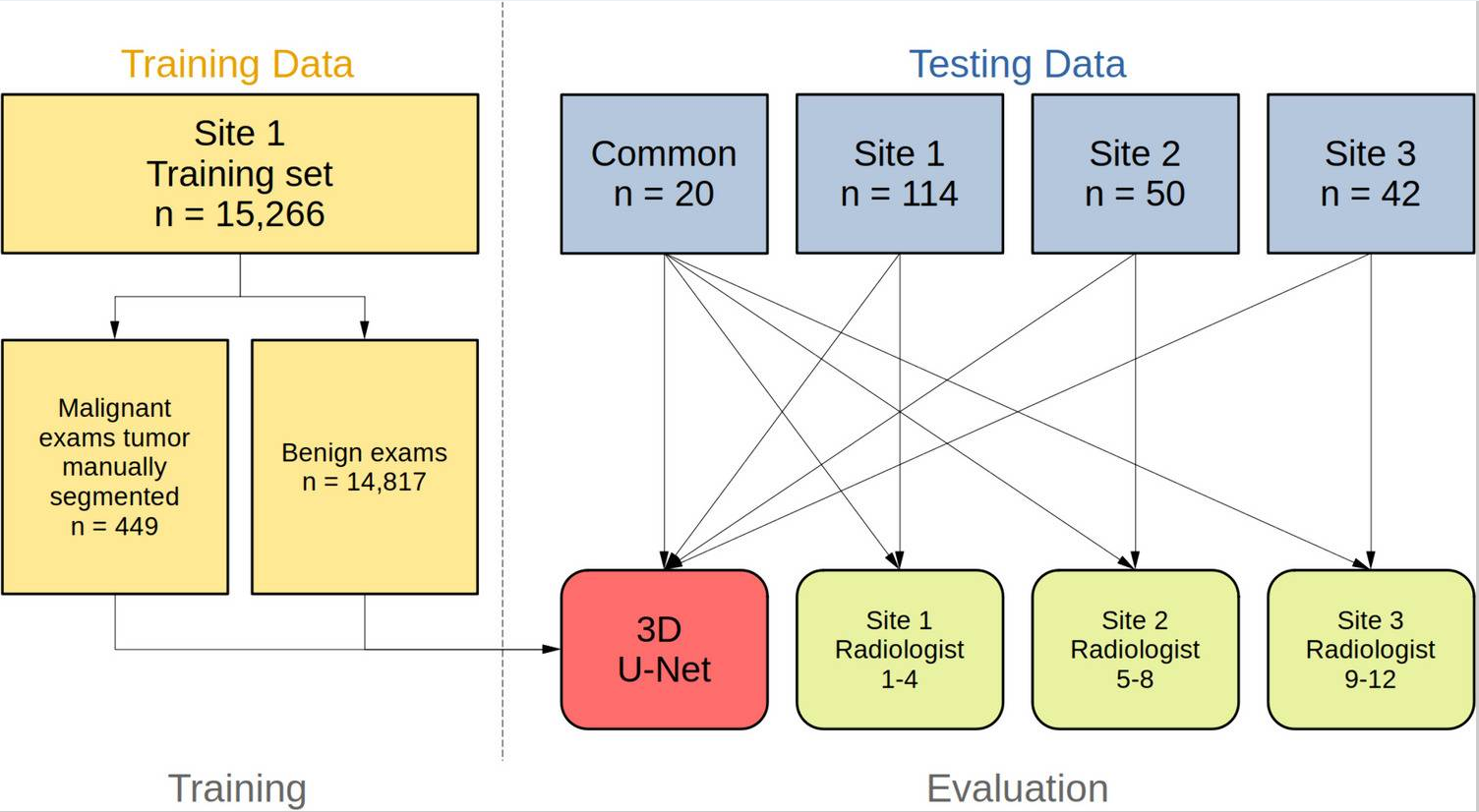 Cross-site Validation of AI Segmentation and Harmonization in Breast MRIYu Huang*, Nicholas J. Leotta*, Lukas Hirsch, Roberto Lo Gullo, Mary Hughes, and 12 more authorsJournal of Imaging Informatics in Medicine, Sep 2024
Cross-site Validation of AI Segmentation and Harmonization in Breast MRIYu Huang*, Nicholas J. Leotta*, Lukas Hirsch, Roberto Lo Gullo, Mary Hughes, and 12 more authorsJournal of Imaging Informatics in Medicine, Sep 2024This work aims to perform a cross-site validation of automated segmentation for breast cancers in MRI and to compare the performance to radiologists. A three-dimensional (3D) U-Net was trained to segment cancers in dynamic contrast-enhanced axial MRIs using a large dataset from Site 1 (n = 15,266; 449 malignant and 14,817 benign). Performance was validated on site-specific test data from this and two additional sites, and common publicly available testing data. Four radiologists from each of the three clinical sites provided two-dimensional (2D) segmentations as ground truth. Segmentation performance did not differ between the network and radiologists on the test data from Sites 1 and 2 or the common public data (median Dice score Site 1, network 0.86 vs. radiologist 0.85, n = 114; Site 2, 0.91 vs. 0.91, n = 50; common: 0.93 vs. 0.90). For Site 3, an affine input layer was fine-tuned using segmentation labels, resulting in comparable performance between the network and radiologist (0.88 vs. 0.89, n = 42). Radiologist performance differed on the common test data, and the network numerically outperformed 11 of the 12 radiologists (median Dice: 0.85–0.94, n = 20). In conclusion, a deep network with a novel supervised harmonization technique matches radiologists’ performance in MRI tumor segmentation across clinical sites. We make code and weights publicly available to promote reproducible AI in radiology.
2023
-
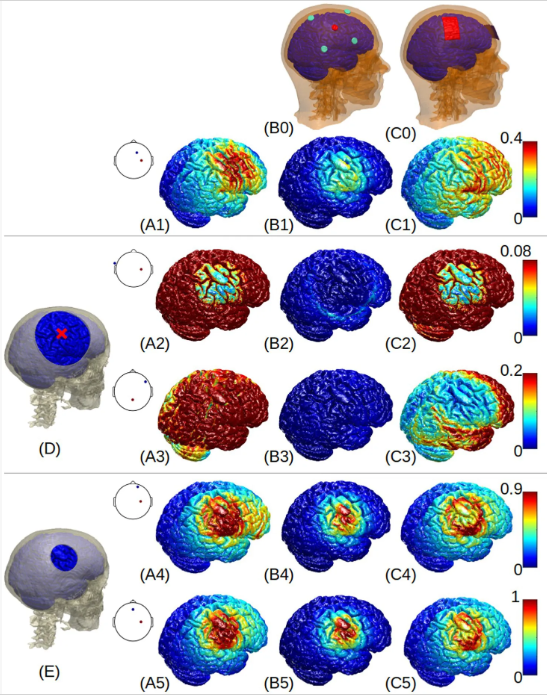 Optimized high-definition tDCS in patients with skull defects and skull platesAlexander Guillen, Dennis Q. Truong, Abhishek Datta, and Yu HuangFrontiers in Human Neuroscience, 2023
Optimized high-definition tDCS in patients with skull defects and skull platesAlexander Guillen, Dennis Q. Truong, Abhishek Datta, and Yu HuangFrontiers in Human Neuroscience, 2023Introduction: Transcranial direct current stimulation (tDCS) has been shown to benefit patients with brain lesions or traumatic brain injury (TBI). These patients usually have skull defects with different sizes and electrical conductivities. There is very little data in the literature that show how to optimally stimulate these patients with the presence of skull defects.Methods: Here we leveraged high-resolution (1 mm) realistic head models to explore the best montages targeting right beneath the skull defects with different sizes and conductivities. Specifically, open-source software ROAST was used to solve for the lead field on the publicly available MIDA model. Four different skull defects/plates were modeled with the center above the right primary motor cortex: a larger defect (10 cm diameter) modeled as either titanium or acrylic plate, and a smaller defect (2.5 cm diameter) modeled as either acute state filled with cerebrospinal fluid (CSF) or chronic state with scar tissue. Optimized stimulation with maximal intensity was run using ROAST targeting the right primary motor cortex.Results: We show that optimized high-definition montages can achieve an average of 0.3 V/m higher stimulation intensities at the target compared to un-optimized montages (M1-SO or 4×1). Large skull defects with titanium or acrylic plates significantly reduce the stimulation intensity by about 80%, while small defects with acute (CSF) or chronic (scar) tissues significantly increase the stimulation intensity by about 200%. Furthermore, one can use M1-SO to achieve almost the same stimulation strength as the optimized montage if the skull has a large defect with titanium plate, and there is no significant difference in stimulation intensity between 4×1 montage and the optimized montage for small skull defects with scar tissue.Discussion: Based on this work, future modeling studies leveraging individual anatomy of skull defects may help guide tDCS practice on patients with skull defects and skull plates.
-
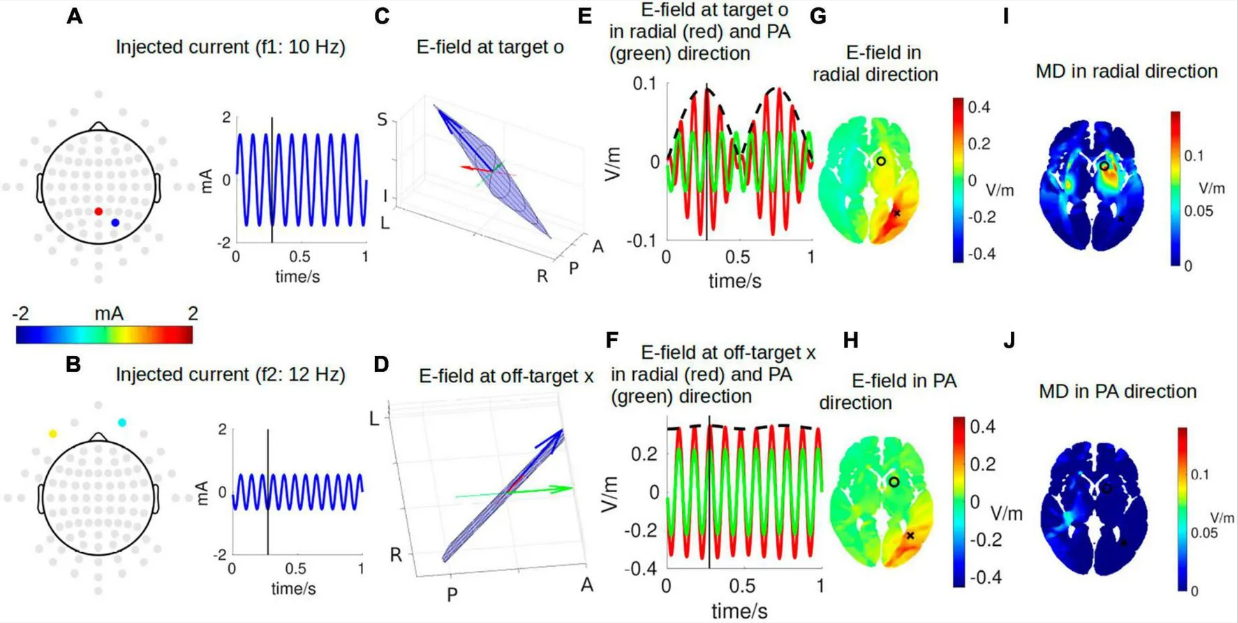 Visualizing interferential stimulation of human brainsYu HuangFrontiers in Human Neuroscience, 2023
Visualizing interferential stimulation of human brainsYu HuangFrontiers in Human Neuroscience, 2023Introduction: Transcranial electrical stimulation (TES) is limited in focally stimulating deep-brain regions, even with optimized stimulation montages. Recently, interferential stimulation (IFS), also known as transcranial temporal interference stimulation (TI, TIS, or tTIS), has drawn much attention in the TES community as both computational and experimental studies show that IFS can reach deep-brain areas. However, the underlying electrodynamics of IFS is complicated and difficult to visualize. Existing literature only shows static visualization of the interfered electric field induced by IFS. These could result in a simplified understanding that there is always one static focal spot between the two pairs of stimulation electrodes. This static visualization can be frequently found in the IFS literature. Here, we aimed to systematically visualize the entire dynamics of IFS.Methods and results: Following the previous study, the lead field was solved for the MNI-152 head, and optimal montages using either two pairs of electrodes or two arrays of electrodes were found to stimulate a deep-brain region close to the left striatum with the highest possible focality. We then visualized the two stimulating electrical currents injected with similar frequencies. We animated the instant electric field vector at the target and one exemplary off-target location both in 3D space and as a 2D Lissajous curve. We finally visualized the distribution of the interfered electric field and the amplitude modulation envelope at an axial slice going through the target location. These two quantities were visualized in two directions: radial-in and posterior–anterior.Discussion: We hope that with intuitive visualization, this study can contribute as an educational resource to the community’s understanding of IFS as a powerful modality for non-invasive focal deep-brain stimulation.
2022
-
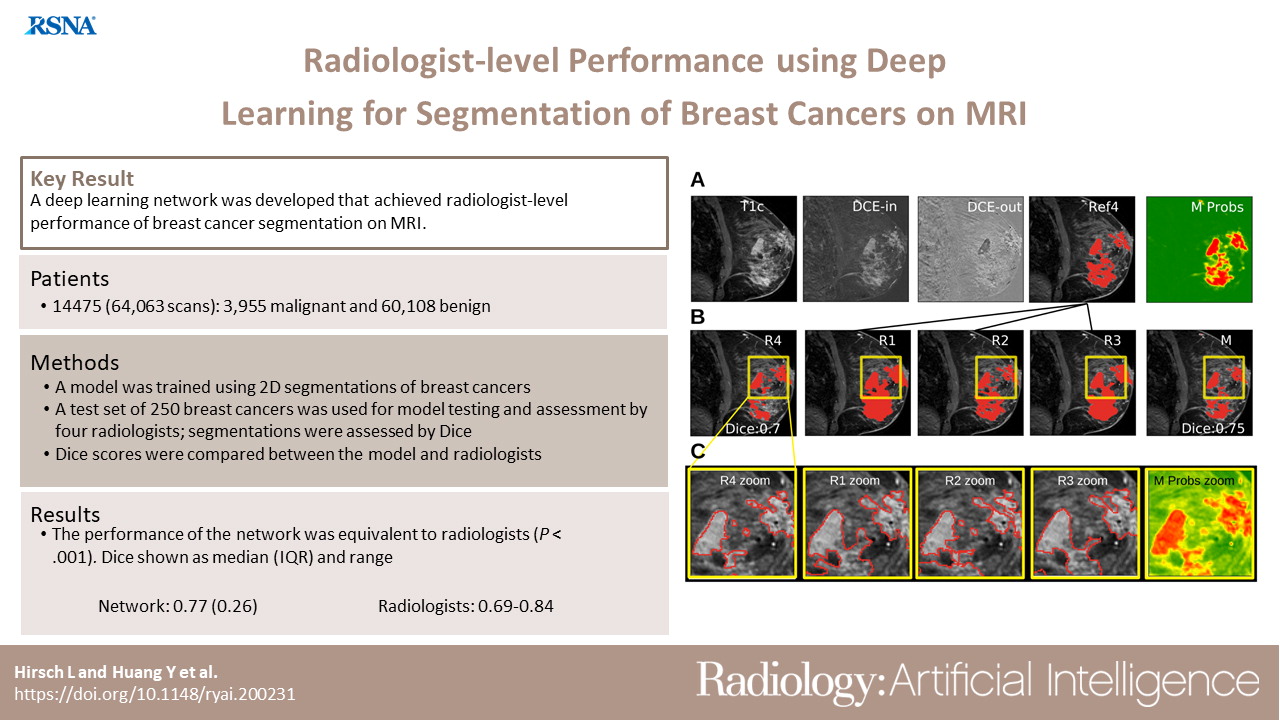 Radiologist-Level Performance by Using Deep Learning for Segmentation of Breast Cancers on MRI ScansLukas Hirsch*, Yu Huang*, Shaojun Luo, Carolina Rossi Saccarelli, Roberto Lo Gullo, and 17 more authorsRadiology: Artificial Intelligence, Jan 2022
Radiologist-Level Performance by Using Deep Learning for Segmentation of Breast Cancers on MRI ScansLukas Hirsch*, Yu Huang*, Shaojun Luo, Carolina Rossi Saccarelli, Roberto Lo Gullo, and 17 more authorsRadiology: Artificial Intelligence, Jan 2022Purpose To develop a deep network architecture that would achieve fully automated radiologist-level segmentation of cancers at breast MRI. Materials and Methods In this retrospective study, 38 229 examinations (composed of 64 063 individual breast scans from 14 475 patients) were performed in female patients (age range, 12–94 years; mean age, 52 years ± 10 [standard deviation]) who presented between 2002 and 2014 at a single clinical site. A total of 2555 breast cancers were selected that had been segmented on two-dimensional (2D) images by radiologists, as well as 60 108 benign breasts that served as examples of noncancerous tissue; all these were used for model training. For testing, an additional 250 breast cancers were segmented independently on 2D images by four radiologists. Authors selected among several three-dimensional (3D) deep convolutional neural network architectures, input modalities, and harmonization methods. The outcome measure was the Dice score for 2D segmentation, which was compared between the network and radiologists by using the Wilcoxon signed rank test and the two one-sided test procedure. Results The highest-performing network on the training set was a 3D U-Net with dynamic contrast-enhanced MRI as input and with intensity normalized for each examination. In the test set, the median Dice score of this network was 0.77 (interquartile range, 0.26). The performance of the network was equivalent to that of the radiologists (two one-sided test procedures with radiologist performance of 0.69–0.84 as equivalence bounds, P \textless .001 for both; n = 250). Conclusion When trained on a sufficiently large dataset, the developed 3D U-Net performed as well as fellowship-trained radiologists in detailed 2D segmentation of breast cancers at routine clinical MRI.
-
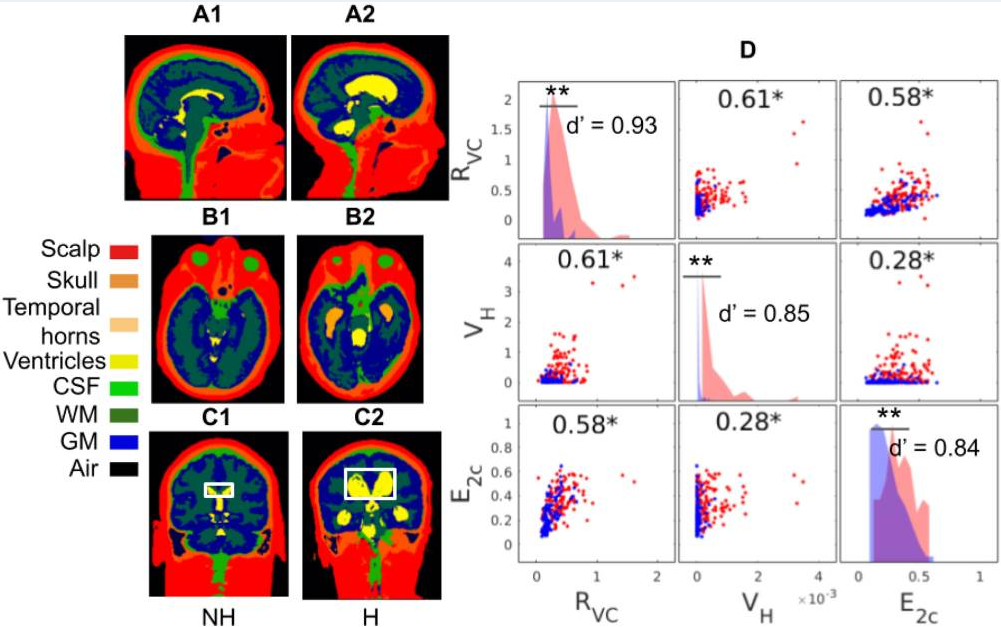 Deep Learning Achieves Neuroradiologist-Level Performance in Detecting Hydrocephalus Requiring TreatmentYu Huang*, Raquel Moreno*, Rachna Malani, Alicia Meng, Nathaniel Swinburne, and 7 more authorsJournal of Digital Imaging, May 2022
Deep Learning Achieves Neuroradiologist-Level Performance in Detecting Hydrocephalus Requiring TreatmentYu Huang*, Raquel Moreno*, Rachna Malani, Alicia Meng, Nathaniel Swinburne, and 7 more authorsJournal of Digital Imaging, May 2022In large clinical centers a small subset of patients present with hydrocephalus that requires surgical treatment. We aimed to develop a screening tool to detect such cases from the head MRI with performance comparable to neuroradiologists. We leveraged 496 clinical MRI exams collected retrospectively at a single clinical site from patients referred for any reason. This diagnostic dataset was enriched to have 259 hydrocephalus cases. A 3D convolutional neural network was trained on 16 manually segmented exams (ten hydrocephalus) and subsequently used to automatically segment the remaining 480 exams and extract volumetric anatomical features. A linear classifier of these features was trained on 240 exams to detect cases of hydrocephalus that required treatment with surgical intervention. Performance was compared to four neuroradiologists on the remaining 240 exams. Performance was also evaluated on a separate screening dataset of 451 exams collected from a routine clinical population to predict the consensus reading from four neuroradiologists using images alone. The pipeline was also tested on an external dataset of 31 exams from a 2nd clinical site. The most discriminant features were the Magnetic Resonance Hydrocephalic Index (MRHI), ventricle volume, and the ratio between ventricle and brain volume. At matching sensitivity, the specificity of the machine and the neuroradiologists did not show significant differences for detection of hydrocephalus on either dataset (proportions test, p \textgreater 0.05). ROC performance compared favorably with the state-of-the-art (AUC 0.90–0.96), and replicated in the external validation. Hydrocephalus cases requiring treatment can be detected automatically from MRI in a heterogeneous patient population based on quantitative characterization of brain anatomy with performance comparable to that of neuroradiologists.
2021
-
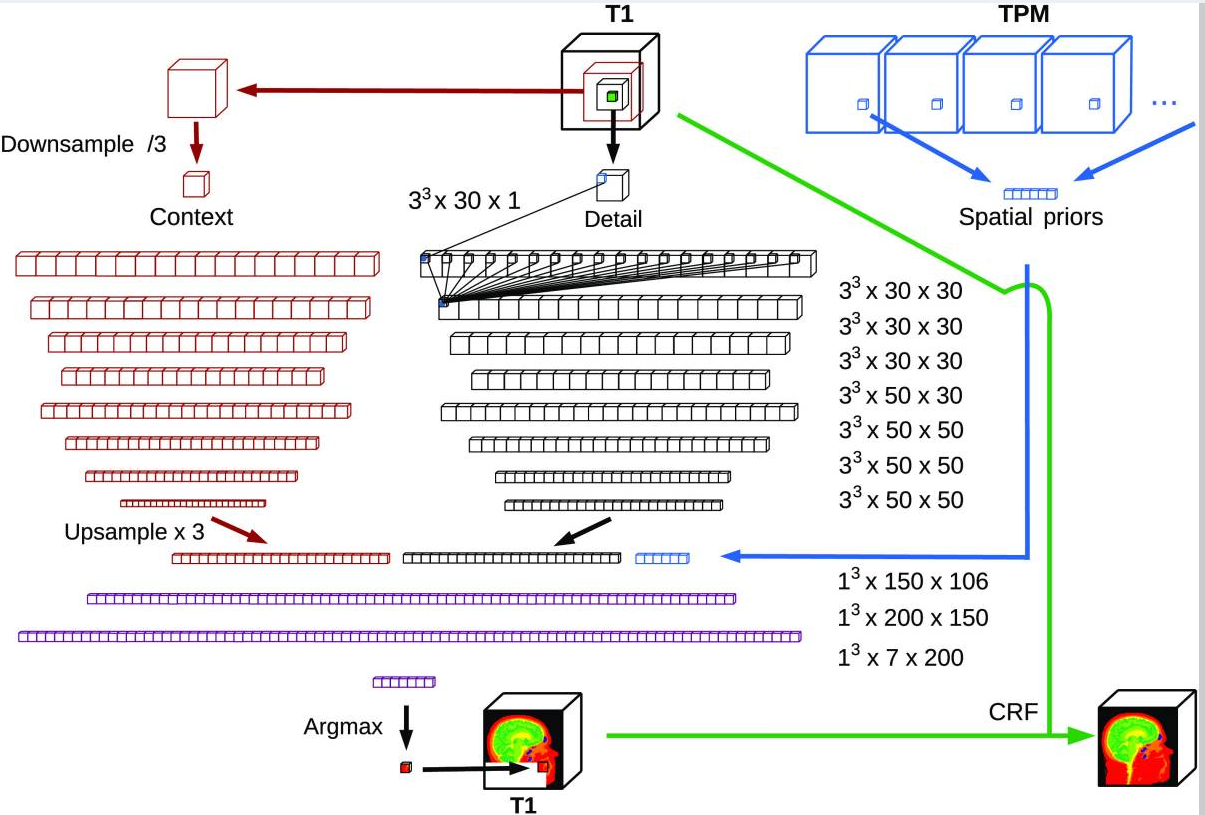 Segmentation of MRI head anatomy using deep volumetric networks and multiple spatial priorsLukas Hirsch, Yu Huang*, and Lucas C. Parra*Journal of Medical Imaging, Jun 2021
Segmentation of MRI head anatomy using deep volumetric networks and multiple spatial priorsLukas Hirsch, Yu Huang*, and Lucas C. Parra*Journal of Medical Imaging, Jun 2021Purpose: Conventional automated segmentation of the head anatomy in magnetic resonance images distinguishes different brain and nonbrain tissues based on image intensities and prior tissue probability maps (TPMs). This works well for normal head anatomies but fails in the presence of unexpected lesions. Deep convolutional neural networks (CNNs) leverage instead spatial patterns and can learn to segment lesions but often ignore prior probabilities. Approach: We add three sources of prior information to a three-dimensional (3D) convolutional network, namely, spatial priors with a TPM, morphological priors with conditional random fields, and spatial context with a wider field-of-view at lower resolution. We train and test these networks on 3D images of 43 stroke patients and 4 healthy individuals which have been manually segmented. Results: We demonstrate the benefits of each source of prior information, and we show that the new architecture, which we call Multiprior network, improves the performance of existing segmentation software, such as SPM, FSL, and DeepMedic for abnormal anatomies. The relevance of the different priors was compared, and the TPM was found to be most beneficial. The benefit of adding a TPM is generic in that it can boost the performance of established segmentation networks such as the DeepMedic and a UNet. We also provide an out-of-sample validation and clinical application of the approach on an additional 47 patients with disorders of consciousness. We make the code and trained networks freely available. Conclusions: Biomedical images follow imaging protocols that can be leveraged as prior information into deep CNNs to improve performance. The network segmentations match human manual corrections performed in 3D and are comparable in performance to human segmentations obtained from scratch in 2D for abnormal brain anatomies.
-
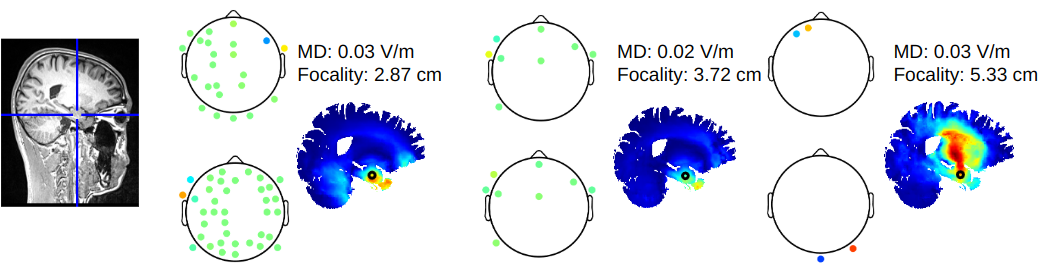 Comparison of optimized interferential stimulation using two pairs of electrodes and two arrays of electrodesYu Huang and Abhishek DattaIn 2021 43rd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Nov 2021
Comparison of optimized interferential stimulation using two pairs of electrodes and two arrays of electrodesYu Huang and Abhishek DattaIn 2021 43rd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Nov 2021Interferential stimulation (IFS) or Temporal Interference (TI) has recently generated considerable interest as computational models show that it can focally stimulate deep brain regions with non-invasive transcranial electrical currents [1]. However, the proposed solution in [1] requires two arrays, involving dozens of electrodes in each, to achieve optimal focality in the deep brain regions. Implementation of this approach is usually not feasible in practice due to the limited number of channels and the associated accuracy and precision needed in current stimulation devices. Alternative method [2] focuses on using only two pairs of electrodes as proposed in the conventional IFS approach and searching exhaustively in the parameter space for the optimal montage that maximizes the focality of the modulation at the deep target. Here we compare these two methods in terms of the quality of the solutions (focality versus modulation depth) and the practicality (speed and number of electrodes needed). We then give general guidelines for optimal IFS in practice for future studies.
2020
-
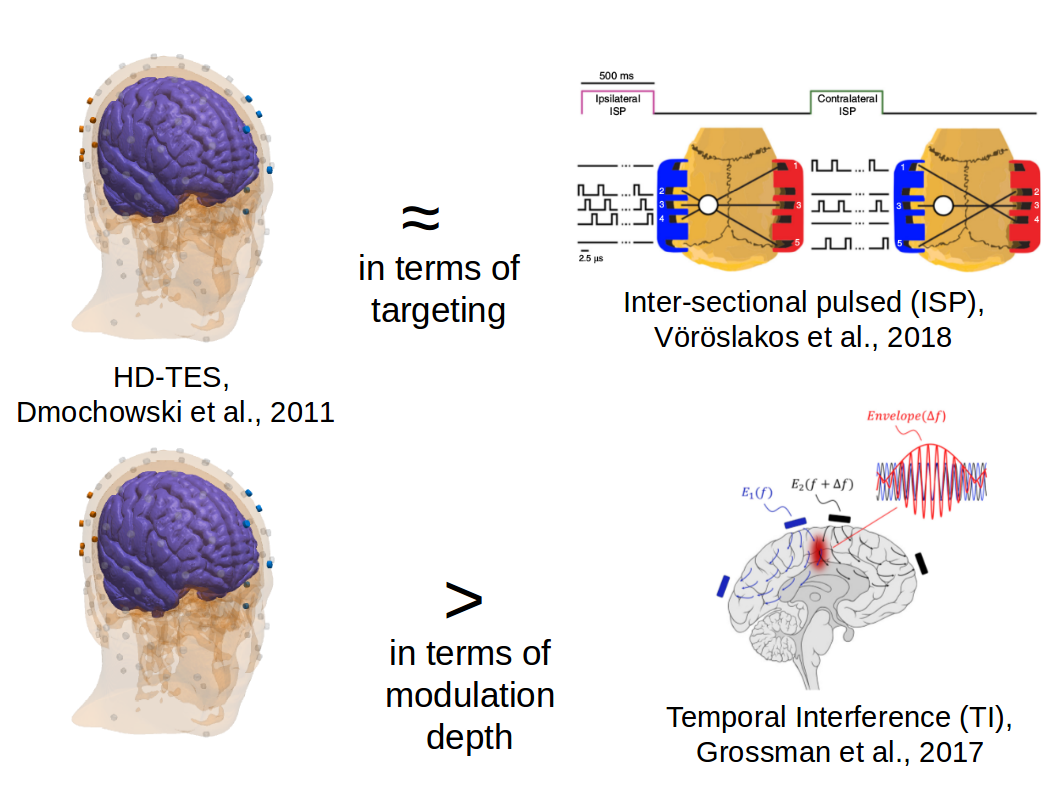
 Optimization of interferential stimulation of the human brain with electrode arraysYu Huang, Abhishek Datta, and Lucas C. ParraJournal of Neural Engineering, Jun 2020
Optimization of interferential stimulation of the human brain with electrode arraysYu Huang, Abhishek Datta, and Lucas C. ParraJournal of Neural Engineering, Jun 2020Objective. Interferential stimulation (IFS) has generated considerable interest recently because of its potential to achieve focal electric fields in deep brain areas with transcranial currents. Conventionally, IFS applies sinusoidal currents through two electrode pairs with close-by frequencies. Here we propose to use an array of electrodes instead of just two electrode pairs; and to use algorithmic optimization to identify the currents required at each electrode to target a desired location in the brain. Approach. We formulate rigorous optimization criteria for IFS to achieve either maximal modulation-depth or maximally focal stimulation. We find the solution for optimal modulation-depth analytically and maximize for focal stimulation numerically. Main results. Maximal modulation is achieved when IFS equals conventional high-definition multi-electrode transcranial electrical stimulation (HD-TES) with a modulated current source. This optimal solution can be found directly from a current-flow model, i.e. the ‘lead field’ without the need for algorithmic optimization. Once currents are optimized numerically to achieve optimal focal stimulation, we find that IFS can indeed be more focal than conventional HD-TES, both at the cortical surface and deep in the brain. Generally, however, stimulation intensity of IFS is weak and the locus of highest intensity does not match the locus of highest modulation. Significance. This proof-of-principle study shows the potential of IFS over HD-TES for focal non-invasive deep brain stimulation. Future work will be needed to improve on intensity of stimulation and convergence of the optimization procedure.
2019
-
 Realistic volumetric-approach to simulate transcranial electric stimulation—ROAST—a fully automated open-source pipelineYu Huang, Abhishek Datta, Marom Bikson, and Lucas C. ParraJournal of Neural Engineering, Jul 2019
Realistic volumetric-approach to simulate transcranial electric stimulation—ROAST—a fully automated open-source pipelineYu Huang, Abhishek Datta, Marom Bikson, and Lucas C. ParraJournal of Neural Engineering, Jul 2019Objective. Research in the area of transcranial electrical stimulation (TES) often relies on computational models of current flow in the brain. Models are built based on magnetic resonance images (MRI) of the human head to capture detailed individual anatomy. To simulate current flow on an individual, the subject’s MRI is segmented, virtual electrodes are placed on this anatomical model, the volume is tessellated into a mesh, and a finite element model (FEM) is solved numerically to estimate the current flow. Various software tools are available for each of these steps, as well as processing pipelines that connect these tools for automated or semi-automated processing. The goal of the present tool—realistic volumetric-approach to simulate transcranial electric simulation (ROAST)—is to provide an end-to-end pipeline that can automatically process individual heads with realistic volumetric anatomy leveraging open-source software and custom scripts to improve segmentation and execute electrode placement. Approach. ROAST combines the segmentation algorithm of SPM12, a Matlab script for touch-up and automatic electrode placement, the finite element mesher iso2mesh and the solver getDP. We compared its performance with commercial FEM software, and SimNIBS, a well-established open-source modeling pipeline. Main results. The electric fields estimated with ROAST differ little from the results obtained with commercial meshing and FEM solving software. We also do not find large differences between the various automated segmentation methods used by ROAST and SimNIBS. We do find bigger differences when volumetric segmentation are converted into surfaces in SimNIBS. However, evaluation on intracranial recordings from human subjects suggests that ROAST and SimNIBS are not significantly different in predicting field distribution, provided that users have detailed knowledge of SimNIBS. Significance. We hope that the detailed comparisons presented here of various choices in this modeling pipeline can provide guidance for future tool development. We released ROAST as an open-source, easy-to-install and fully-automated pipeline for individualized TES modeling.
-
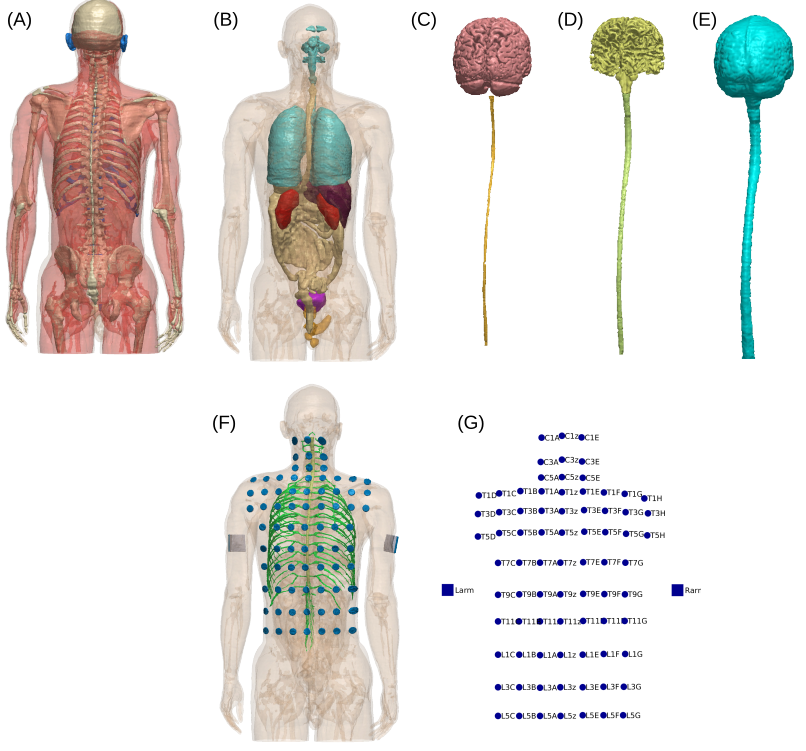 Optimized Transcutaneous Spinal Cord Direct Current Stimulation using Multiple Electrodes from 3/9/7 SystemYu Huang, Chris Thomas, and Abhishek DattaIn 2019 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Jul 2019
Optimized Transcutaneous Spinal Cord Direct Current Stimulation using Multiple Electrodes from 3/9/7 SystemYu Huang, Chris Thomas, and Abhishek DattaIn 2019 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Jul 2019Transcutaneous spinal cord direct current stimulation (tSDCS) has been applied as an easy non-invasive approach to modulate spinal cord functions. Currently there is no formal layout or guidelines for electrode placement to optimize tSDCS. Most clinical applications simply place the stimulating electrode over the intended spinal cord target. Here we show that this ad hoc method cannot achieve optimal stimulation. Specifically, we propose a new electrode layout for optimized tSDCS. The candidate high-definition electrodes distribute on the back of the body evenly and the layout was named 3/9/7 system. Algorithmic optimization was performed leveraging this electrode placement system and a 1 mm3 human full body model. Results show that the optimal stimulation montages cannot be trivially determined and they outperform the unoptimized stimulation configuration. This work opens the possibility for systematic treatment planning in future clinical applications of tSDCS.
-
 Can transcranial electric stimulation with multiple electrodes reach deep targets?Yu Huang and Lucas C. ParraBrain Stimulation, Jan 2019
Can transcranial electric stimulation with multiple electrodes reach deep targets?Yu Huang and Lucas C. ParraBrain Stimulation, Jan 2019To reach a deep target in the brain with transcranial electric stimulation (TES), currents have to pass also through the cortical surface. Thus, it is generally thought that TES cannot achieve focal deep brain stimulation. Recent efforts with interfering waveforms and pulsed stimulation have argued that one can achieve deeper or more intense stimulation in the brain. Here we argue that conventional transcranial stimulation with multiple current sources is just as effective as these new approaches. The conventional multi-electrode approach can be numerically optimized to maximize intensity or focality at a desired target location. Using such optimal electrode configurations we find in a detailed and realistic head model that deep targets may in fact be strongly stimulated, with cerebro-spinal fluid guiding currents deep into the brain.
2018
-
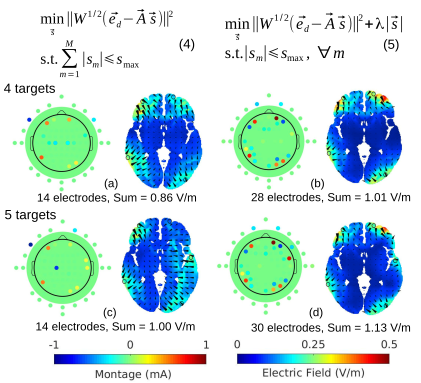 Optimized tDCS for Targeting Multiple Brain Regions: An Integrated ImplementationYu Huang, Chris Thomas, Abhishek Datta, and Lucas C. ParraIn 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Jul 2018
Optimized tDCS for Targeting Multiple Brain Regions: An Integrated ImplementationYu Huang, Chris Thomas, Abhishek Datta, and Lucas C. ParraIn 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Jul 2018Transcranial direct current stimulation (tDCS) aims to deliver weak electric current into the brain to modulate neural activities. Based on the volume conductor model of the head, optimization algorithm can be used to determine a specific montage of high-definition electrodes on the scalp to achieve targeted stimulation. However, simultaneous targeting for multiple disconnected regions can rarely be found in the literature. Here we attempted to provide an integrated solution for optimized tDCS to target multiple brain regions (either a single point or brain structures). By improving the “maxintensity” routine previously published in [1], we are able to target two regions of interest (ROI) in the brain simultaneously.For ROIs more than two, we show that the “max-focality” algorithm using weighted least-square in [1] can be further improved by putting the L1-norm constraint on the stimulation current as apenalty term into the cost function. Up to five ROIs can be targeted at the same time without violating the safety criteria. Further analysis shows that, for multiple targets, a trade-off exists between targeting accuracy and the number of electrodes needed. We implemented all these algorithms in Soterix software HD-TargetsTM.
2017
-
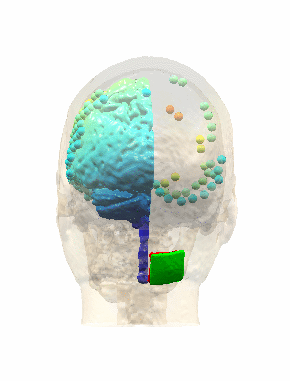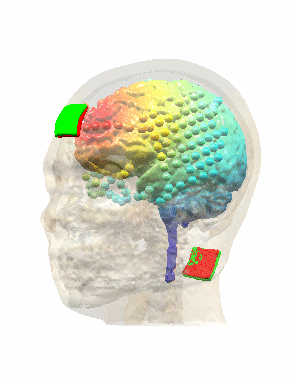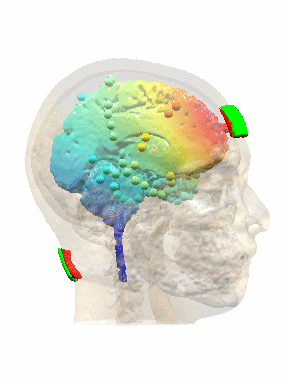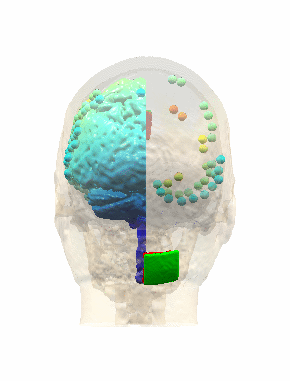 Measurements and models of electric fields in the in vivo human brain during transcranial electric stimulationYu Huang*, Anli A. Liu*, Belen Lafon, Daniel Friedman, Michael Dayan, and 5 more authorseLife, Feb 2017
Measurements and models of electric fields in the in vivo human brain during transcranial electric stimulationYu Huang*, Anli A. Liu*, Belen Lafon, Daniel Friedman, Michael Dayan, and 5 more authorseLife, Feb 2017Transcranial electric stimulation aims to stimulate the brain by applying weak electrical currents at the scalp. However, the magnitude and spatial distribution of electric fields in the human brain are unknown. We measured electric potentials intracranially in ten epilepsy patients and estimate electric fields across the entire brain by leveraging calibrated current-flow models. When stimulating at 2 mA, cortical electric fields reach 0.4 V/m, the lower limit of effectiveness in animal studies. When individual whole-head anatomy is considered, the predicted electric field magnitudes correlate with the recorded values in cortical (r=0.89) and depth (r=0.84) electrodes. Accurate models require adjustment of tissue conductivity values reported in the literature, but accuracy is not improved when incorporating white matter anisotropy or different skull compartments. This is the first study to validate and calibrate current-flow models with in vivo intracranial recordings in humans, providing a solid foundation to target stimulation and interpret clinical trials.
2016
-
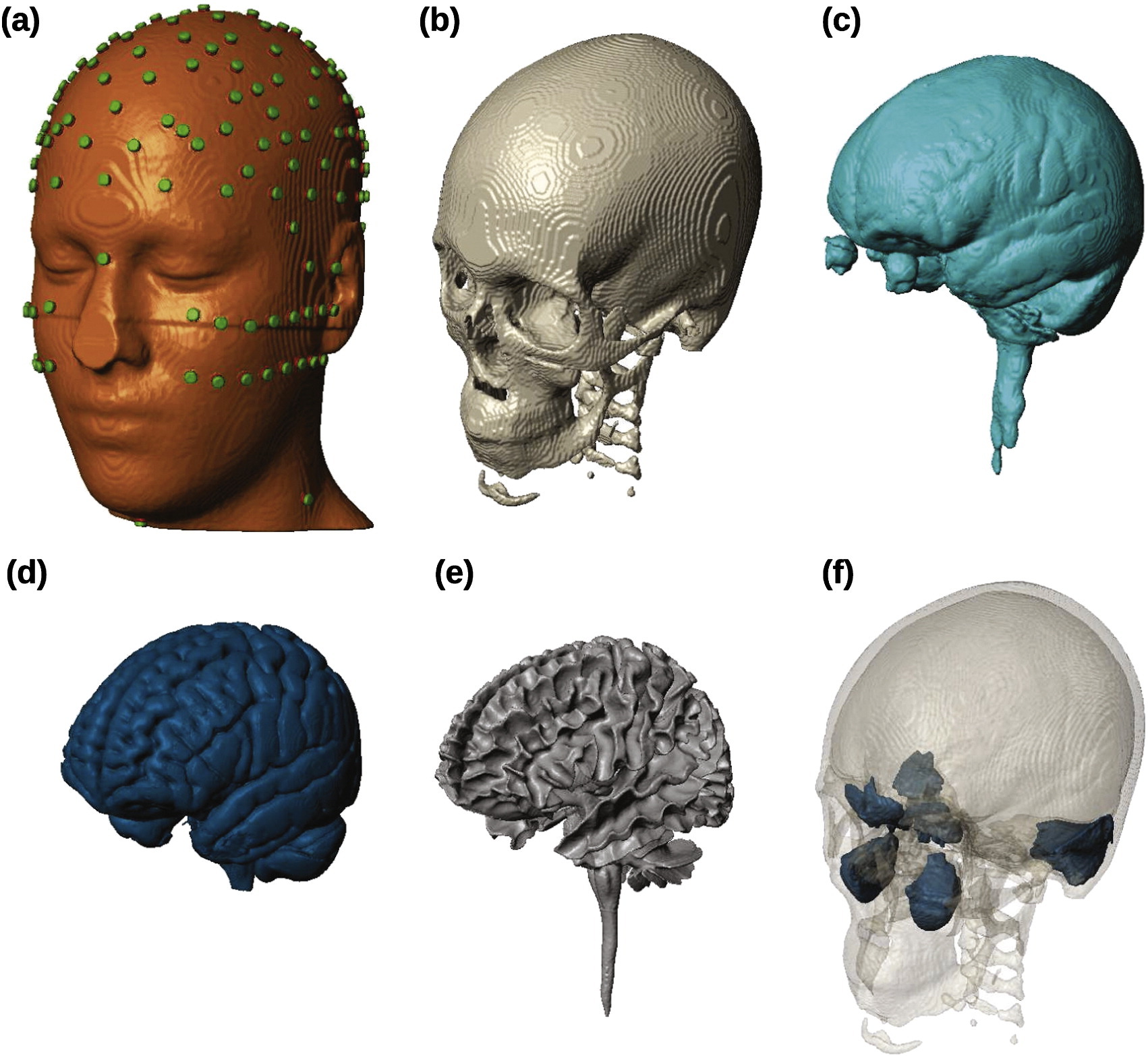 The New York Head—A precise standardized volume conductor model for EEG source localization and tES targetingYu Huang, Lucas C. Parra, and Stefan HaufeNeuroImage, Oct 2016
The New York Head—A precise standardized volume conductor model for EEG source localization and tES targetingYu Huang, Lucas C. Parra, and Stefan HaufeNeuroImage, Oct 2016In source localization of electroencephalograpic (EEG) signals, as well as in targeted transcranial electric current stimulation (tES), a volume conductor model is required to describe the flow of electric currents in the head. Boundary element models (BEM) can be readily computed to represent major tissue compartments, but cannot encode detailed anatomical information within compartments. Finite element models (FEM) can capture more tissue types and intricate anatomical structures, but with the higher precision also comes the need for semi-automated segmentation, and a higher computational cost. In either case, adjusting to the individual human anatomy requires costly magnetic resonance imaging (MRI), and thus head modeling is often based on the anatomy of an ‘arbitrary’ individual (e.g. Colin27). Additionally, existing reference models for the human head often do not include the cerebro-spinal fluid (CSF), and their field of view excludes portions of the head and neck—two factors that demonstrably affect current-flow patterns. Here we present a highly detailed FEM, which we call ICBM-NY, or "New York Head". It is based on the ICBM152 anatomical template (a non-linear average of the MRI of 152 adult human brains) defined in MNI coordinates, for which we extended the field of view to the neck and performed a detailed segmentation of six tissue types (scalp, skull, CSF, gray matter, white matter, air cavities) at 0.5 mm 3 resolution. The model was solved for 231 electrode locations. To evaluate its performance, additional FEMs and BEMs were constructed for four individual subjects. Each of the four individual FEMs (regarded as the ‘ground truth’) is compared to its BEM counterpart, the ICBM-NY, a BEM of the ICBM anatomy, an ‘individualized’ BEM of the ICBM anatomy warped to the individual head surface, and FEMs of the other individuals. Performance is measured in terms of EEG source localization and tES targeting errors. Results show that the ICBM-NY outperforms FEMs of mismatched individual anatomies as well as the BEM of the ICBM anatomy according to both criteria. We therefore propose the New York Head as a new standard head model to be used in future EEG and tES studies whenever an individual MRI is not available. We release all model data online at neuralengr.com/nyhead/ to facilitate broad adoption.
2015
-
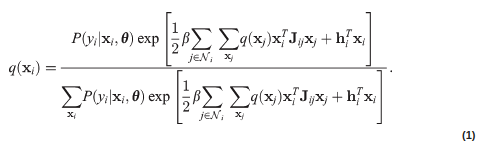 Fully Automated Whole-Head Segmentation with Improved Smoothness and Continuity, with Theory ReviewedYu Huang and Lucas C. ParraPLOS ONE, May 2015
Fully Automated Whole-Head Segmentation with Improved Smoothness and Continuity, with Theory ReviewedYu Huang and Lucas C. ParraPLOS ONE, May 2015Individualized current-flow models are needed for precise targeting of brain structures using transcranial electrical or magnetic stimulation (TES/TMS). The same is true for current-source reconstruction in electroencephalography and magnetoencephalography (EEG/MEG). The first step in generating such models is to obtain an accurate segmentation of individual head anatomy, including not only brain but also cerebrospinal fluid (CSF), skull and soft tissues, with a field of view (FOV) that covers the whole head. Currently available automated segmentation tools only provide results for brain tissues, have a limited FOV, and do not guarantee continuity and smoothness of tissues, which is crucially important for accurate current-flow estimates. Here we present a tool that addresses these needs. It is based on a rigorous Bayesian inference framework that combines image intensity model, anatomical prior (atlas) and morphological constraints using Markov random fields (MRF). The method is evaluated on 20 simulated and 8 real head volumes acquired with magnetic resonance imaging (MRI) at 1 mm3 resolution. We find improved surface smoothness and continuity as compared to the segmentation algorithms currently implemented in Statistical Parametric Mapping (SPM). With this tool, accurate and morphologically correct modeling of the whole-head anatomy for individual subjects may now be feasible on a routine basis. Code and data are fully integrated into SPM software tool and are made publicly available. In addition, a review on the MRI segmentation using atlas and the MRF over the last 20 years is also provided, with the general mathematical framework clearly derived.
2013
-
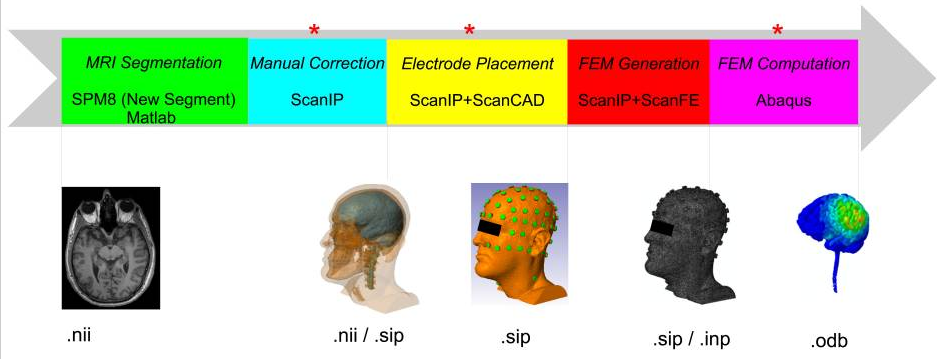 Automated MRI segmentation for individualized modeling of current flow in the human headYu Huang, Jacek P. Dmochowski, Yuzhuo Su, Abhishek Datta, Christopher Rorden, and 1 more authorJournal of Neural Engineering, 2013
Automated MRI segmentation for individualized modeling of current flow in the human headYu Huang, Jacek P. Dmochowski, Yuzhuo Su, Abhishek Datta, Christopher Rorden, and 1 more authorJournal of Neural Engineering, 2013Objective. High-definition transcranial direct current stimulation (HD-tDCS) and high-density electroencephalography require accurate models of current flow for precise targeting and current source reconstruction. At a minimum, such modeling must capture the idiosyncratic anatomy of the brain, cerebrospinal fluid (CSF) and skull for each individual subject. Currently, the process to build such high-resolution individualized models from structural magnetic resonance images requires labor-intensive manual segmentation, even when utilizing available automated segmentation tools. Also, accurate placement of many high-density electrodes on an individual scalp is a tedious procedure. The goal was to develop fully automated techniques to reduce the manual effort in such a modeling process. Approach. A fully automated segmentation technique based on Statical Parametric Mapping 8, including an improved tissue probability map and an automated correction routine for segmentation errors, was developed, along with an automated electrode placement tool for high-density arrays. The performance of these automated routines was evaluated against results from manual segmentation on four healthy subjects and seven stroke patients. The criteria include segmentation accuracy, the difference of current flow distributions in resulting HD-tDCS models and the optimized current flow intensities on cortical targets. Main results. The segmentation tool can segment out not just the brain but also provide accurate results for CSF, skull and other soft tissues with a field of view extending to the neck. Compared to manual results, automated segmentation deviates by only 7% and 18% for normal and stroke subjects, respectively. The predicted electric fields in the brain deviate by 12% and 29% respectively, which is well within the variability observed for various modeling choices. Finally, optimized current flow intensities on cortical targets do not differ significantly. Significance. Fully automated individualized modeling may now be feasible for large-sample EEG research studies and tDCS clinical trials.
2009
-
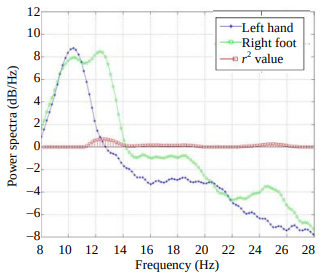 An Algorithm for Idle-State Detection and Continuous Classifier Design in Motor-Imagery-Based BCIYu Huang, Qiang Wu, Xu Lei, Ping Yang, Peng Xu, and 1 more authorJournal of Electronic Science and Technology, Mar 2009
An Algorithm for Idle-State Detection and Continuous Classifier Design in Motor-Imagery-Based BCIYu Huang, Qiang Wu, Xu Lei, Ping Yang, Peng Xu, and 1 more authorJournal of Electronic Science and Technology, Mar 2009The development of asynchronous braincomputer interface (BCI) based on motor imagery (MI) poses the research in algorithms for detecting the nontask states (i.e., idle state) and the design of continuous classifiers that classify continuously incoming electroencephalogram (EEG) samples. An algorithm is proposed in this paper which integrates two two-class classifiers to detect idle state and utilizes a sliding window to achieve continuous outputs. The common spatial pattern (CSP) algorithm is used to extract features of EEG signals and the linear support vector machine (SVM) is utilized to serve as classifier. The algorithm is applied on dataset IVb of BCI competition III, with a resulting mean square error of 0.66. The result indicates that the proposed algorithm is feasible in the first step of the development of asynchronous systems.
















